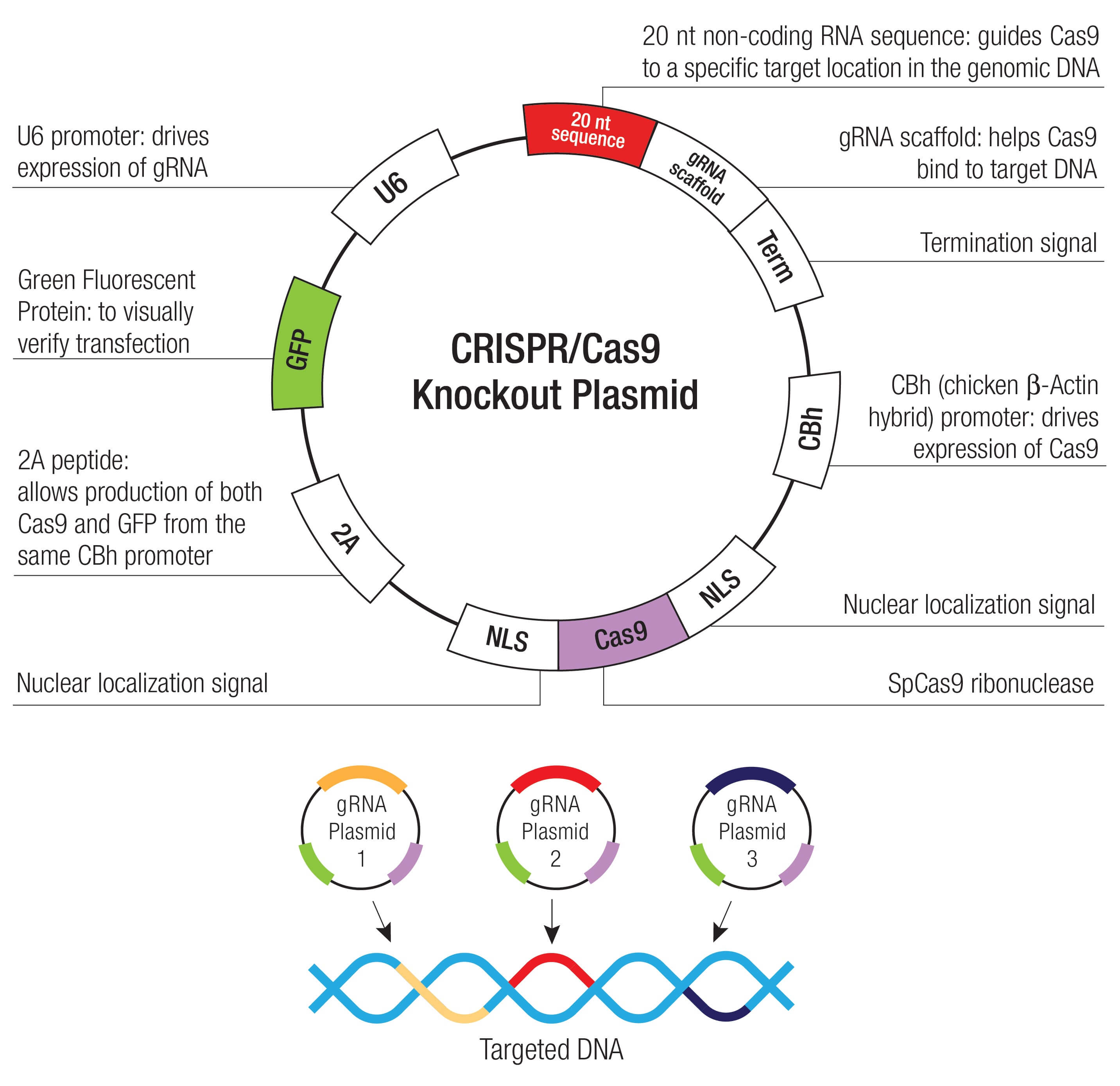
Review: Insights into molecular mechanisms of disease in neurodegeneration with brain iron accumulation: unifying theories - Arber - 2016 - Neuropathology and Applied Neurobiology - Wiley Online Library
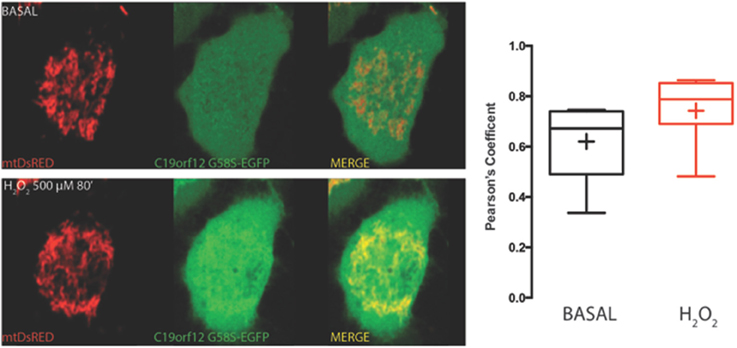
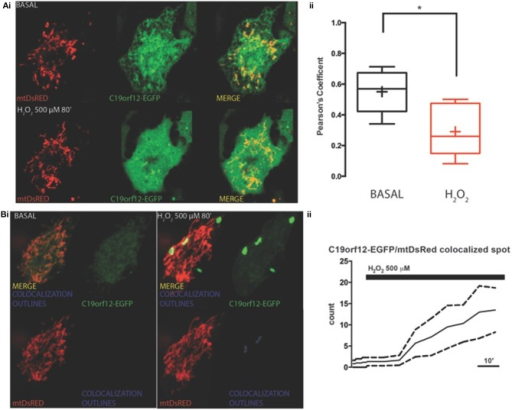
Frontiers | Mutations of C19orf12, coding for a transmembrane glycine zipper containing mitochondrial protein, cause mis-localization of the protein, inability to respond to oxidative stress and increased mitochondrial Ca2+

Nazo, the Drosophila homolog of the NBIA-mutated protein – c19orf12, is required for triglyceride homeostasis | bioRxiv

Pharmaceuticals | Free Full-Text | Neurodegeneration with Brain Iron Accumulation Disorders: Valuable Models Aimed at Understanding the Pathogenesis of Iron Deposition
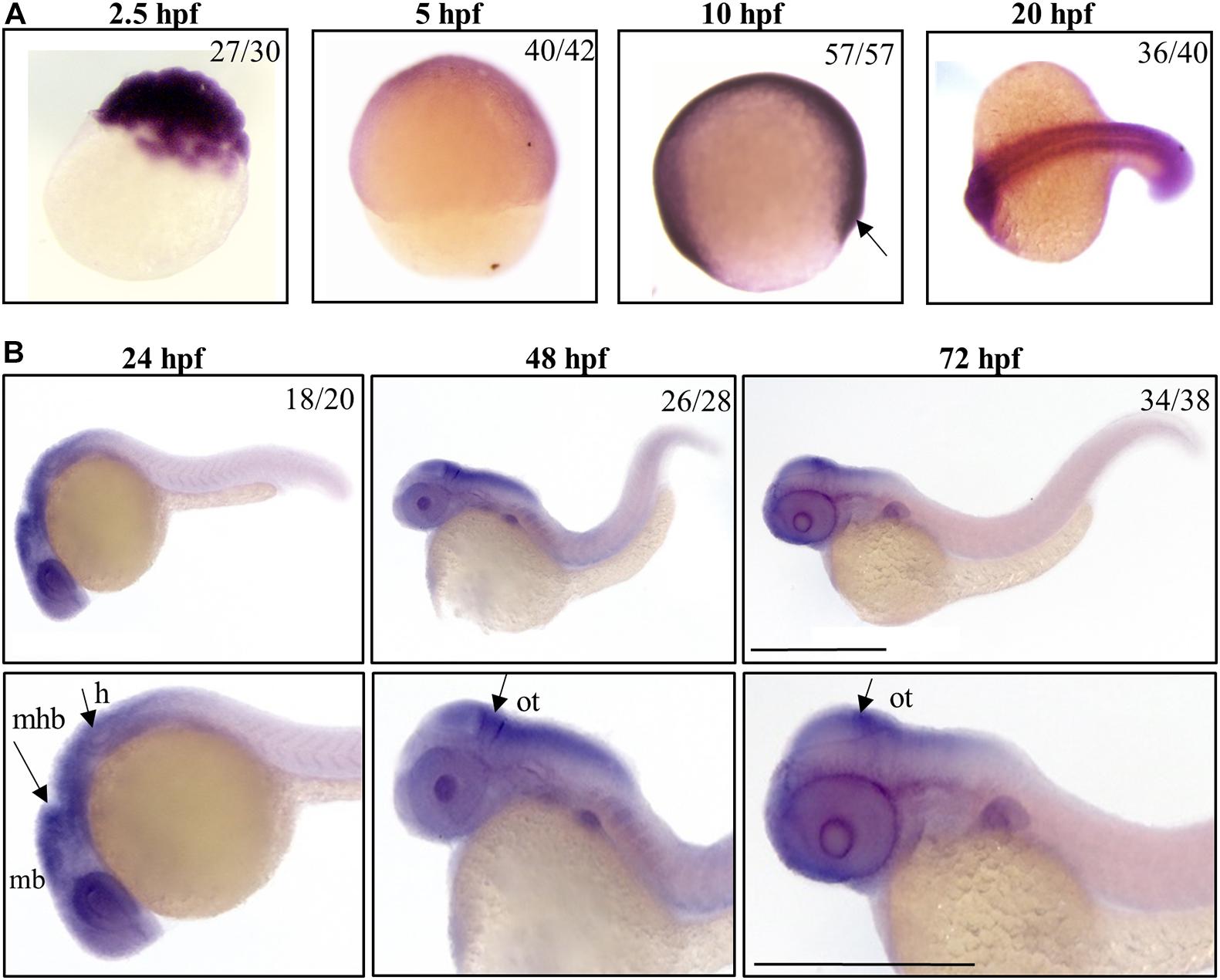
Frontiers | The Downregulation of c19orf12 Negatively Affects Neuronal and Musculature Development in Zebrafish Embryos
Alignment of the fly C19orf12 orthologs (CG3740, CG11671) with human... | Download Scientific Diagram

Absence of an Orphan Mitochondrial Protein, C19orf12, Causes a Distinct Clinical Subtype of Neurodegeneration with Brain Iron Accumulation - ScienceDirect

Dominant mitochondrial membrane protein-associated neurodegeneration (MPAN) variants cluster within a specific C19orf12 isoform - ScienceDirect

Pharmaceutics | Free Full-Text | Identification of Autophagy as a Functional Target Suitable for the Pharmacological Treatment of Mitochondrial Membrane Protein-Associated Neurodegeneration (MPAN) In Vitro
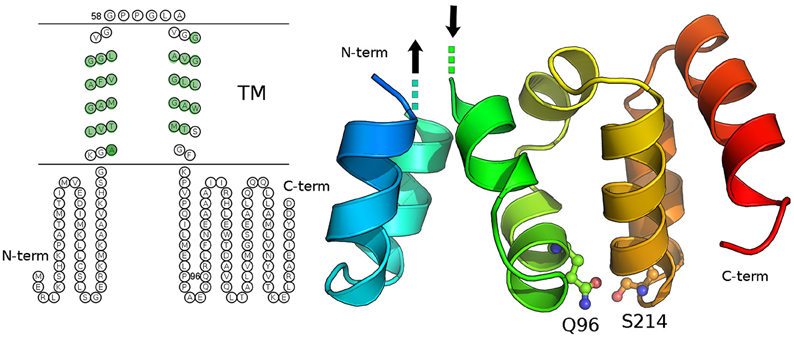
Frontiers | Mutations of C19orf12, coding for a transmembrane glycine zipper containing mitochondrial protein, cause mis-localization of the protein, inability to respond to oxidative stress and increased mitochondrial Ca2+

Brain iron and metabolic abnormalities in C19orf12 mutation carriers: A 7.0 tesla MRI study in mitochondrial membrane protein-associated neurodegeneration. - Munich Cluster for Systems Neurology - LMU Munich
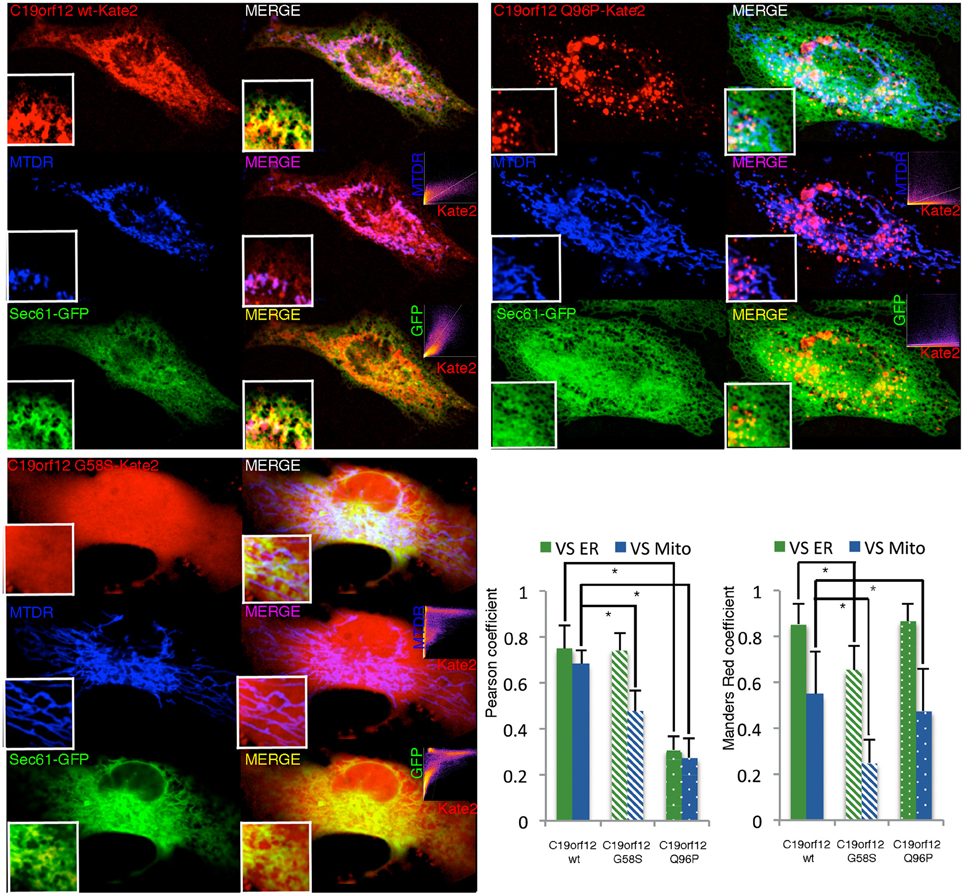
Frontiers | Mutations of C19orf12, coding for a transmembrane glycine zipper containing mitochondrial protein, cause mis-localization of the protein, inability to respond to oxidative stress and increased mitochondrial Ca2+
![PDF] Mutations of C19orf12, coding for a transmembrane glycine zipper containing mitochondrial protein, cause mis-localization of the protein, inability to respond to oxidative stress and increased mitochondrial Ca2+ | Semantic Scholar PDF] Mutations of C19orf12, coding for a transmembrane glycine zipper containing mitochondrial protein, cause mis-localization of the protein, inability to respond to oxidative stress and increased mitochondrial Ca2+ | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/dd2ecb2012d86f9d3853c6aef367791e936d0787/11-Figure9-1.png)
PDF] Mutations of C19orf12, coding for a transmembrane glycine zipper containing mitochondrial protein, cause mis-localization of the protein, inability to respond to oxidative stress and increased mitochondrial Ca2+ | Semantic Scholar

C19orf12 gene mutations in patients with neurodegeneration with brain iron accumulation - ScienceDirect

C19orf12 gene mutations in patients with neurodegeneration with brain iron accumulation - ScienceDirect
![PDF] Mutations of C19orf12, coding for a transmembrane glycine zipper containing mitochondrial protein, cause mis-localization of the protein, inability to respond to oxidative stress and increased mitochondrial Ca2+ | Semantic Scholar PDF] Mutations of C19orf12, coding for a transmembrane glycine zipper containing mitochondrial protein, cause mis-localization of the protein, inability to respond to oxidative stress and increased mitochondrial Ca2+ | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/dd2ecb2012d86f9d3853c6aef367791e936d0787/5-Figure1-1.png)
PDF] Mutations of C19orf12, coding for a transmembrane glycine zipper containing mitochondrial protein, cause mis-localization of the protein, inability to respond to oxidative stress and increased mitochondrial Ca2+ | Semantic Scholar
![PDF] Absence of an orphan mitochondrial protein, c19orf12, causes a distinct clinical subtype of neurodegeneration with brain iron accumulation. | Semantic Scholar PDF] Absence of an orphan mitochondrial protein, c19orf12, causes a distinct clinical subtype of neurodegeneration with brain iron accumulation. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/8f4b65f35621e9f879833a58183ba96b06f5ec7f/4-Figure2-1.png)