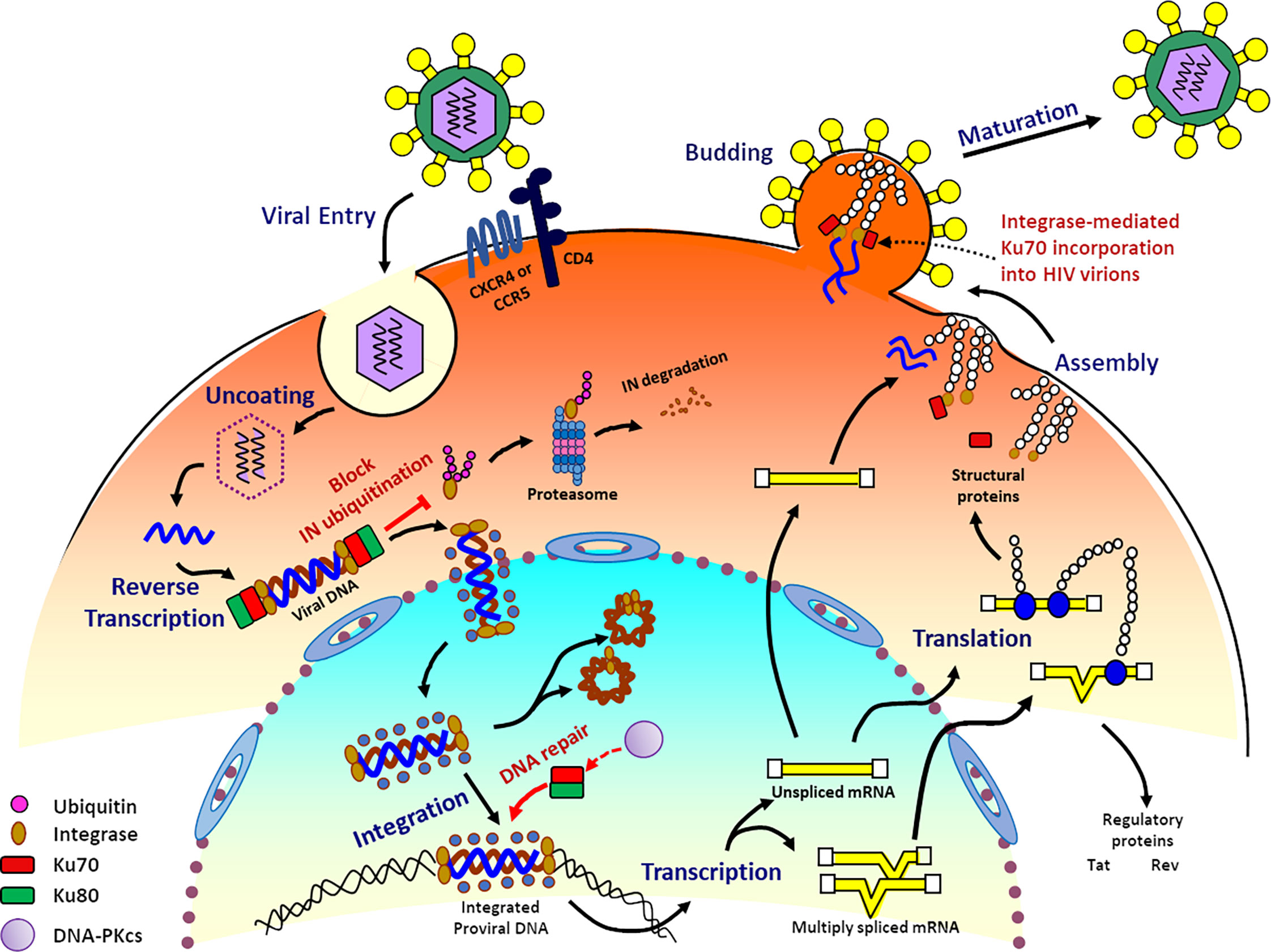
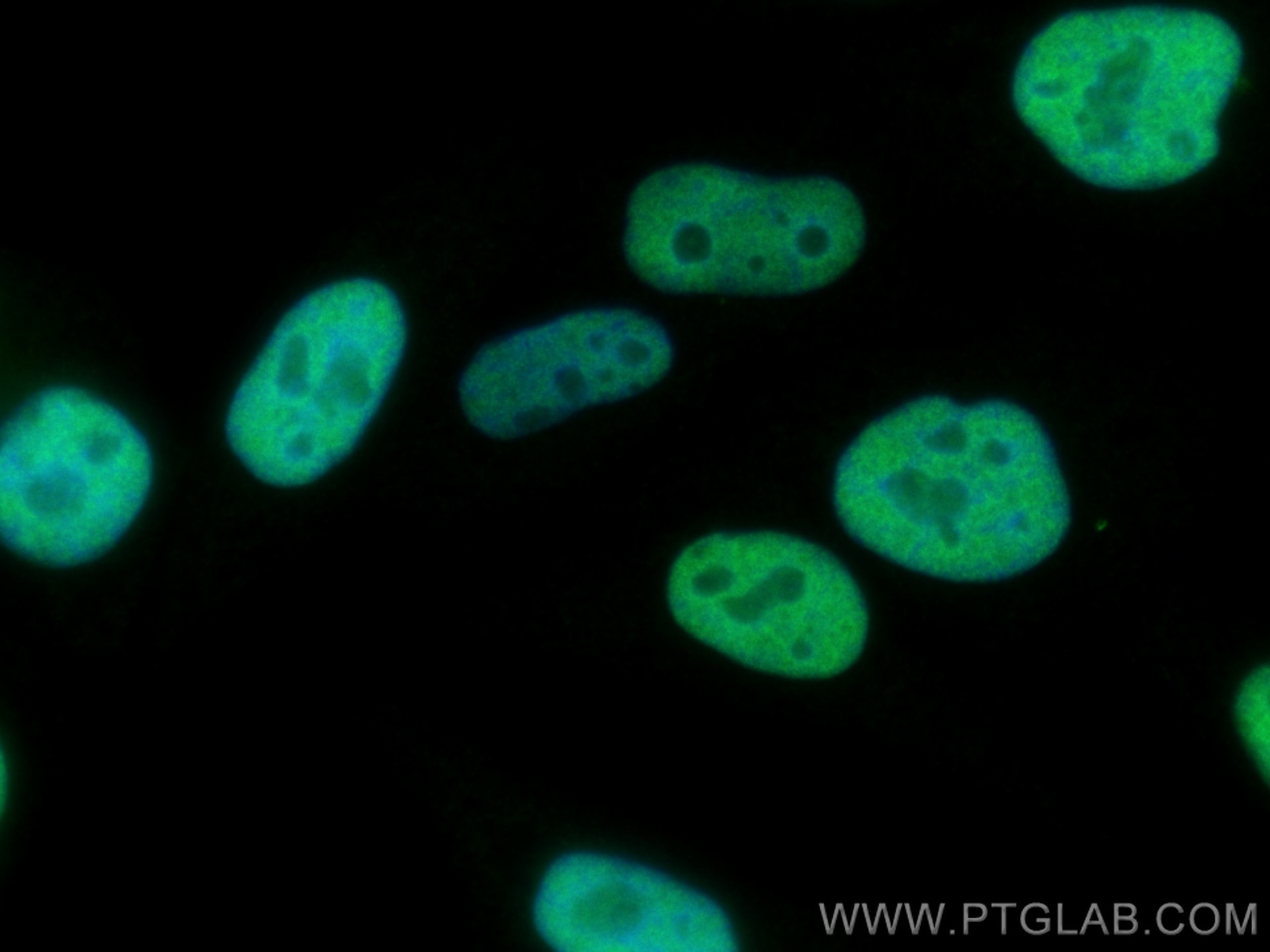
Acetylation of nuclear localization signal controls importin-mediated nuclear transport of Ku70 | bioRxiv

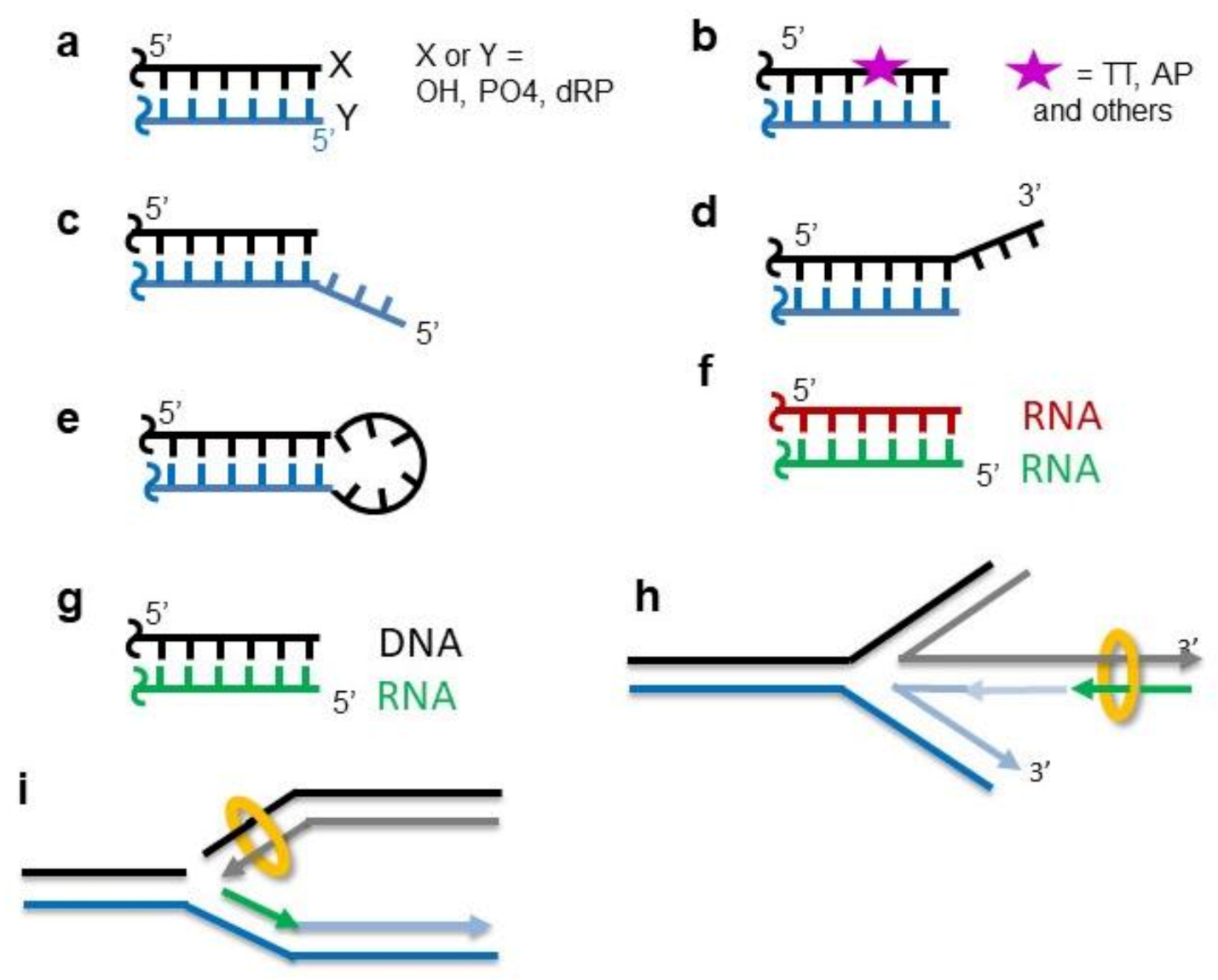
SAP domain forms a flexible part of DNA aperture in Ku70/80 - Hnízda - 2021 - The FEBS Journal - Wiley Online Library

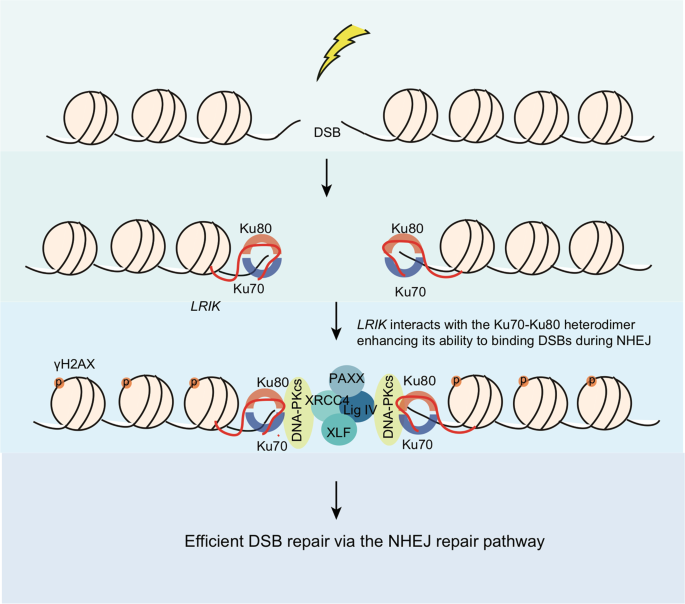
Schematic representation of DSBs repair by NHEJ. Ku70/Ku80 heterodimers... | Download Scientific Diagram

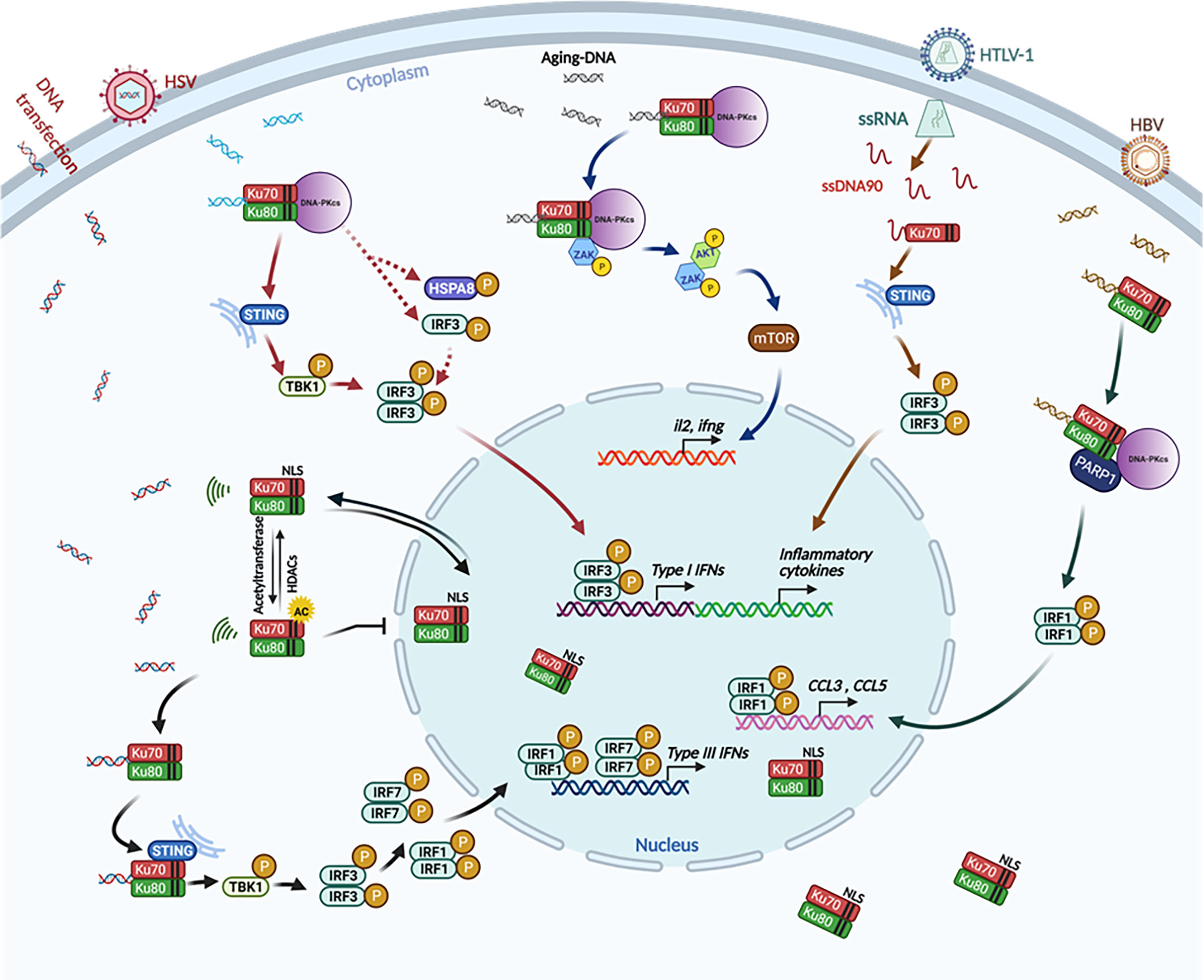
Cytoplasmic‐translocated Ku70 senses intracellular DNA and mediates interferon‐lambda1 induction - Sui - 2021 - Immunology - Wiley Online Library

Non-homologous end joining repair of DSBs. Ku70/80 binds to DNA ends... | Download Scientific Diagram

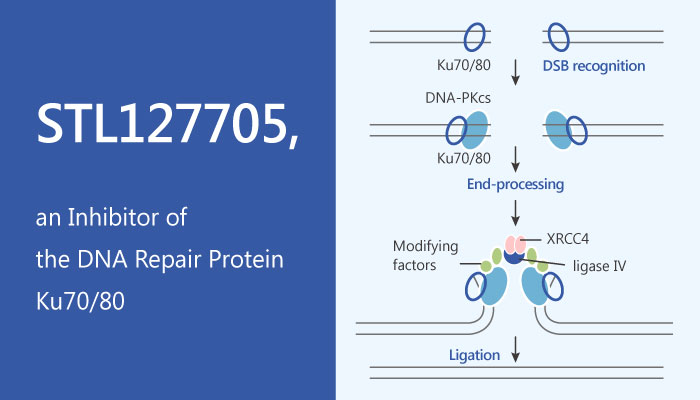
STING is an essential mediator of the Ku70-mediated production of IFN-λ1 in response to exogenous DNA | Science Signaling
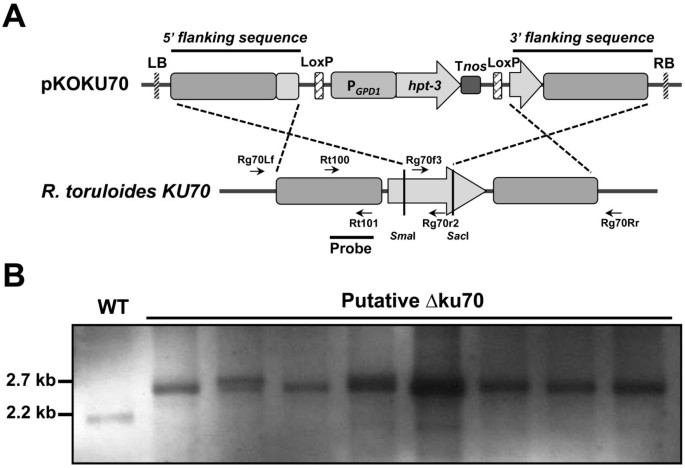
Molecular characterization of KU70 and KU80 homologues and exploitation of a KU70-deficient mutant for improving gene deletion frequency in Rhodosporidium toruloides | BMC Microbiology | Full Text
![PDF] The Ku70/80 ring in Non-Homologous End-Joining: easy to slip on, hard to remove. | Semantic Scholar PDF] The Ku70/80 ring in Non-Homologous End-Joining: easy to slip on, hard to remove. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/0a85ce278521192474569a6c65831c62081866f5/3-Figure2-1.png)
PDF] The Ku70/80 ring in Non-Homologous End-Joining: easy to slip on, hard to remove. | Semantic Scholar

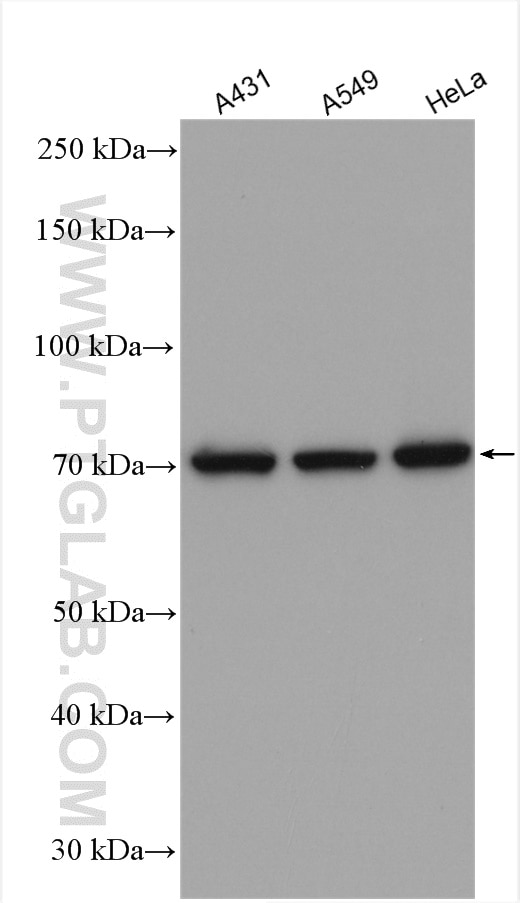
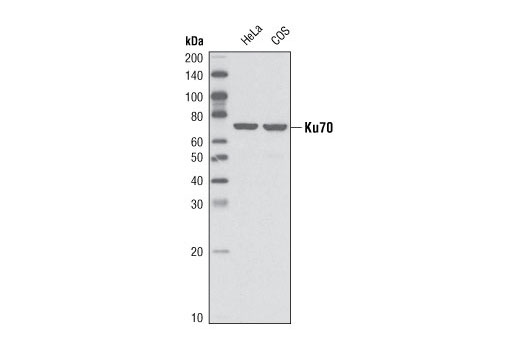

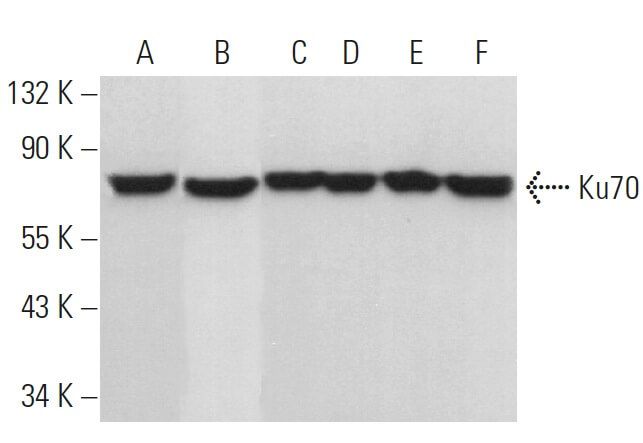
![Recombinant Anti-Ku70 antibody [EPR4027] (ab92450) | Abcam Recombinant Anti-Ku70 antibody [EPR4027] (ab92450) | Abcam](https://www.abcam.com/ps/products/92/ab92450/Images/ab92450-365431-anti-ku70-antibody-epr4027-westernblot-human.jpg)